Miss Casillo
Welcome to 6th grade


Law of Conservation of Energy
The first law of thermodynamics is also called the law of conservation of energy. It states that energy can be transferred from one system to another, but it cannot be destroyed. This is important for example as in the study of climate change. Remember in our last unit on light we learned about how light behaves with black versus white objects. Lets apply this knowledge to soil. Dark soils (closer to black) absorb much more solar energy (storing in the form of heat) than do snow and ice (whiter), which reflect solar radiation. The energy stored in the soil radiates out to warm the air, raising air temperatures; it does not just disappear.
Note: The Earth rotating counterclockwise and the Sun heating our land and water (radiant energy) at different times and temperatures is what give us our wind.
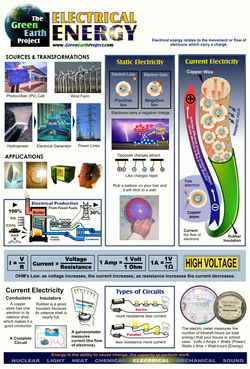
Energy in the form of heat, electricity, motion, or sound can change forms and move from one system to another. This can be demonstrated by making a cup of tea. For example, electricity, one form of energy, changes form to thermal energy (heat) through the stove and is transferred by conduction to the water for tea. The tea warms the cup, which warms the hands of the person holding it. This is energy transfer.
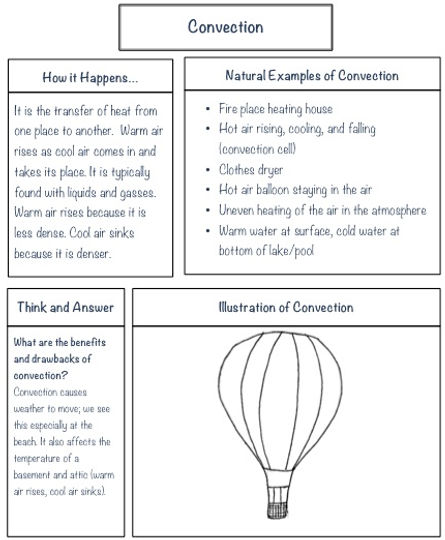
Conduction Convection and Radiation

Definitions of Critical Vocabulary & Underlying Concepts:
Concept/Vocabulary Word Definition Conduction the transfer of heat between two parts of a stationary system, caused by a temperature difference between the parts.
Convection the transfer of heat by the circulation or movement of the heated parts of a liquid or gas.
Energy The ability to do work or cause change.
Radiation the process in which energy is emitted as particles or waves.
Transfer to convey or remove from one place, person, etc., to another:





7 forms of energy: mechanical, sound, chemical, electrical, light, heat and nuclear.
Conservation of Energy
 |  |
|---|---|
 |  |
 |  |
 |  |










